In a high school science class, Jimmy was given 50 millilitres of water and 50 millilitres of ethanol. His task was to mix them together and then run an experiment on the mixture. When the teacher came to check on him, however, Jimmy’s mixture only contained 94 millilitres. The teacher accused him of drinking some of the mixture in an attempt to get intoxicated and immediately sent him to the principal. Jimmy swore that he did not drink the mixture and that he didn’t lose any of the liquids by any means. What happened to the other six millilitres, and how could Jimmy prove his innocence?
Answer
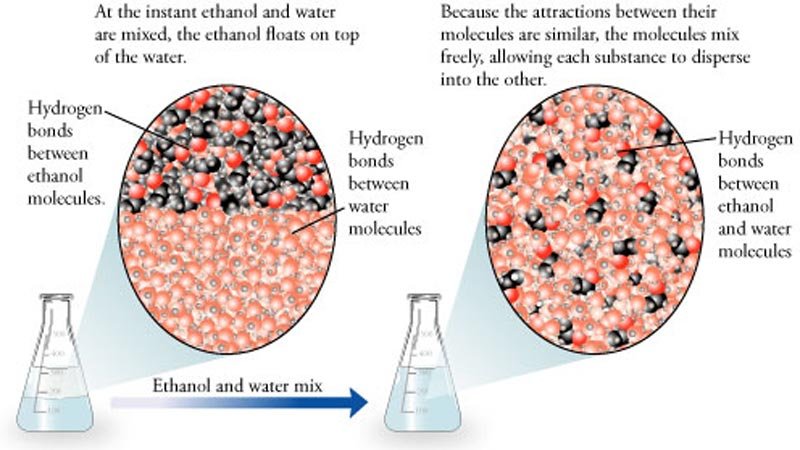
When liquids are mixed, the molecules can arrange themselves so that they may fit together more tightly than either of the original, pure liquids. A good way to visualize this is to picture a box completely filled with billiard balls (representing large molecules). If you pour a small amount of sand (representing small molecules) into the box, the sand will fill the voids between the billiard balls. The mass in the box will increase, but the total volume will not change. This is how Jimmy could prove his innocence. If he weighs the mixture, it will equal the original weight of the water plus the weight of the ethanol.
